Description
ADVANCED TECHNOLOGY AC PROGRAMMABLE MICROCURRENT GENERATOR

DEVICE FUNCTION
Independently programmable base and modulating frequency sets
Programmable microcurrent frequency specific parameters
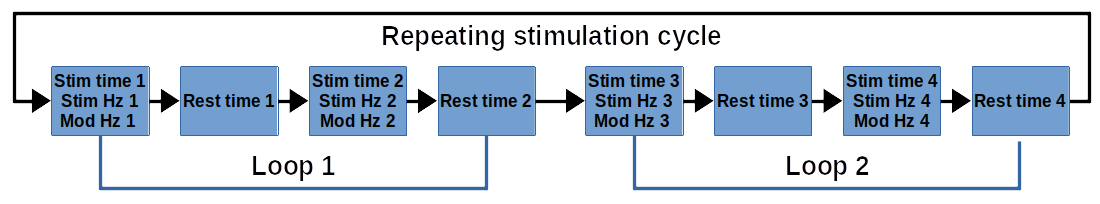
– 4 independent Base Frequencies (1-20,000 Hz)
– 4 independent stimulation times of the 4 Base Frequencies (1-20 minutes)
– 4 independent Amplitude Modulating Frequencies (0-100 Hz) of the 4 Base Frequencies
– 4 independent rest times between the 4 Base Frequency stimulation times (0-20 minutes)
– Loop 2 is independently programmable, can be programmed as duplicate of Loop 1 or can be inactivated.
– Algorithm programmable, constant-current, self adaptive Output Voltage.
– Waveform output parameters remain valid throughout real world impedance values up to 300 kiloohms (compared to insufficient ≤ 10 kiloohms max limit of most devices).
– Waveform shape re-calculated based on programmed variables at beginning of each stimulation loop for precision waveform generation and transmission.
– Smart statistical circuit monitoring and user alerts with on-screen corrective instructions.
– Device logging of electrode contact data for monitoring and analysis, exports into spreadsheet with included PC app.
GENERAL HEALTH AND WELLNESS DEVICE USE
The FG300 programmable microcurrent stimulation technology can be used for the general enhancement of whole body cellular processes.
The FG300 shares electronics technologies from the SIS development medical devices. International Patent Pending Application device & technology.





